By Anpel Team | 23 October 2020 | 2 Comments
The brilliant blue analysis of synthetic colorant analysis has noise interference?
The brilliant blue analysis of synthetic colorant analysis has noise interference? Anpu experiment takes you to reveal the truth!

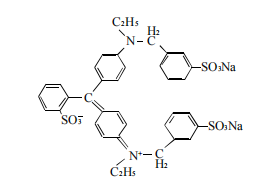
Figure 3 Bright blue structure

Let's uncover the truth together!
1.Machine standard products
Prepare the bright blue standard product and configure it to 10ppm with water.
2.Instrument conditions
Chromatographic column: C18-Wp (4.6mm*250mm, 5um)
Mobile phase: A: methanol B: 0.02M ammonium acetate
Gradient elution: 0min 20%A, 5min 35%A, 12~18min 98%A, 20~27min 20%A
Column temperature: 25℃
Wavelength: 254nm
Injection volume: 20ul
Flow rate: 1.0ml/min
3.Experimental spectrum
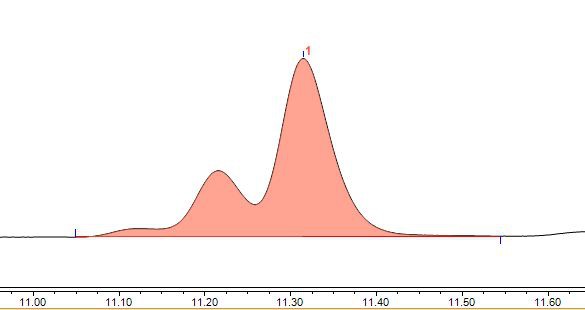
Figure 1 Bright blue spectrum (chromatographic condition 1)
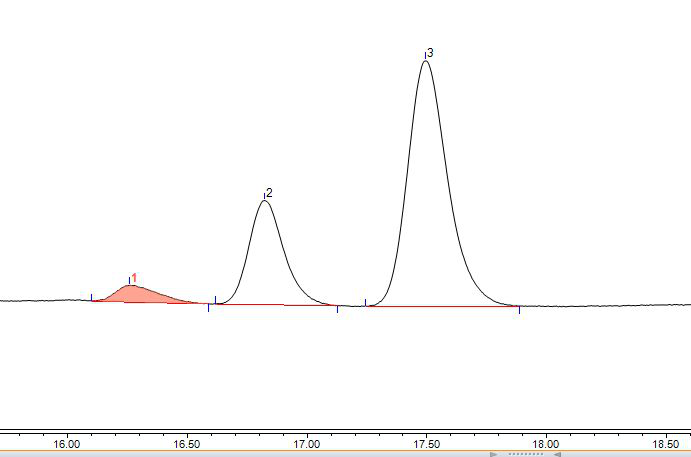
Figure 4 Bright blue spectrum (chromatographic condition 2)
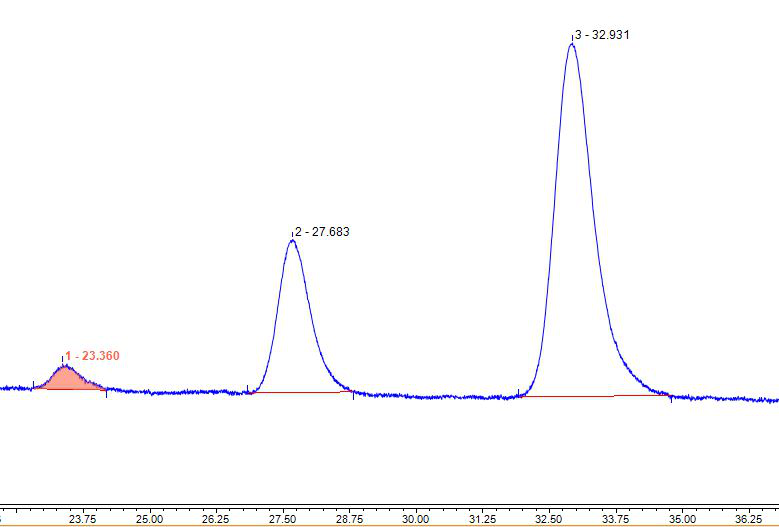
Figure 5 Bright blue spectrum (chromatographic condition 3)

4.Result analysis
The bright blue spectrum obtained after subtracting the solvent blank is shown in Figure 1. It is not a symmetrical chromatographic peak, and not only asymmetric, it looks like three peaks. So what is the reason? We analyzed from the following aspects.
(1) The problem of standard products. If the purity of the standard product is not high, it may cause impurity interference. Check that the purity of the certificate mark of the brilliant blue standa rd product is 87.6%. The purity is not particularly high, but it is produced by a well-known imported brand D company. It is impossible to determine the 3 peaks. Whether it contains impurity peaks.
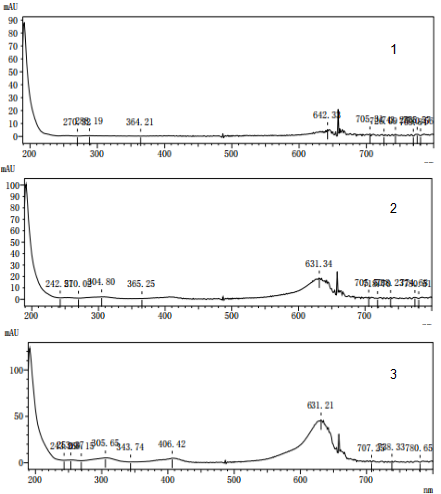
(2) Spectral characteristics. Ultraviolet-visible absorption spectrum is an inherent property of com pounds. Generally, different compounds have different absorption characteristics. Therefore, com pounds can be qualitatively analyzed through spectral characteristics. So what are these three pe aks to the low end? Use a DAD detector to scan them in the range of 200-800nm. The resulting spectrum is shown in Figure 2. It can be seen that the ultraviolet absorption characteristics of the three compounds are very similar. This situation in UV analysis is generally isomers.
1.Machine standard products
Prepare the bright blue standard product and configure it to 10ppm with water.
2.Instrument conditions
Chromatographic column: C18-Wp (4.6mm*250mm, 5um)
Mobile phase: A: methanol B: 0.02M ammonium acetate
Gradient elution: 0min 20%A, 5min 35%A, 12~18min 98%A, 20~27min 20%A
Column temperature: 25℃
Wavelength: 254nm
Injection volume: 20ul
Flow rate: 1.0ml/min
3.Experimental spectrum

Figure 1 Bright blue spectrum (chromatographic condition 1)

Figure 4 Bright blue spectrum (chromatographic condition 2)

Figure 5 Bright blue spectrum (chromatographic condition 3)

4.Result analysis
The bright blue spectrum obtained after subtracting the solvent blank is shown in Figure 1. It is not a symmetrical chromatographic peak, and not only asymmetric, it looks like three peaks. So what is the reason? We analyzed from the following aspects.
(1) The problem of standard products. If the purity of the standard product is not high, it may cause impurity interference. Check that the purity of the certificate mark of the brilliant blue standa rd product is 87.6%. The purity is not particularly high, but it is produced by a well-known imported brand D company. It is impossible to determine the 3 peaks. Whether it contains impurity peaks.
(2) Spectral characteristics. Ultraviolet-visible absorption spectrum is an inherent property of com pounds. Generally, different compounds have different absorption characteristics. Therefore, com pounds can be qualitatively analyzed through spectral characteristics. So what are these three pe aks to the low end? Use a DAD detector to scan them in the range of 200-800nm. The resulting spectrum is shown in Figure 2. It can be seen that the ultraviolet absorption characteristics of the three compounds are very similar. This situation in UV analysis is generally isomers.

Figure 2 Bright blue spectrum scan
(3) Structural features. So does Brilliant Blue really have isomers? The structural formu la of Brilliant Blue is shown in Figure 3. It is a non-azo acid dye. There are mainly 3 isomers. There are often 3 isomers in nature. The mixture (meta-sulfonate: para-sulfo nate: ortho-sulfonate=75~85: 15-20:0~8) exists in the form of a mixture, so a single symmetrical chromatographic peak is often not obtained in liquid phase analysis.

Figure 3 Bright blue structure
(4) Quantitative analysis. So how to quantitatively analyze the content of brilliant blue is a problem that the experimenters are more concerned about. The premise of quanti tative analysis by liquid chromatography is to separate first. Try to adjust the flow ratio to slow down the elution capacity. We found that these three peaks Baseline separ atio n can be achieved, such as Figure 4 and Figure 5. The results of each chromatogra ph ic peak area under different chromatographic conditions and different concentrations ar e shown in Table 1.
Table 1 Bright blue peak area
Table 1 Bright blue peak area

It can be seen that under different chromatographic conditions, there is a certain difference in the peak area of the three chromatographic peaks of the same concentr ation of brilliant blue when the baseline is separated and the peak area is not separat ed. The peak area-concentration linear relationship of the isomers of blue is go od. It may be that the response of bright blue is slightly different under different chro matog raphic conditions, and under specific chromatographic conditions, the The isom ers will not change much.
5.Experimental results
(1) The suspected impurity peaks that appeared in the analysis of brilliant blue are actually the isomers of brilliant blue, and there are 3 isomers of brilliant blue.
5.Experimental results
(1) The suspected impurity peaks that appeared in the analysis of brilliant blue are actually the isomers of brilliant blue, and there are 3 isomers of brilliant blue.
(2) In practical applications, you can decide whether to separate the three isomers completely according to your own experimental purposes. If the total brilliant blue co ntent is tested, combine the three target peaks for calculation, if it is calculated The content of each isomer should be calculated quantitatively after adjusting the chromat ographic conditions to make it completely separated.
Recently Reviews
Read MoreLeave a Reply
Your email address will not be published.Required fields are marked. *
POPULAR BLOG
ARCHIVES
CATEGORIES